In vitro glycoengineering - Suitability for BioPharma manufacturing
Introduction
The importance of Fc glycosylation of monoclonal antibodies (mAbs) with regard to biological activity is widely discussed and has been investigated in numerous studies. It is known that different glycan species contribute differently to the biological activity of therapeutic proteins1. However, even if a certain glycan variant is known to have a positive impact on biological activity, it might not be easy to consistently produce such a glycosylation pattern. Cell line engineering or bioprocess optimization enable to increase glycosylation homogeneity or to at least partially achieve a certain glycan pattern. However, these activities are time-consuming and might compromise other key parameters, such as antibody yield. In addition, it is not economical to produce mAbs
with a certain glycan pattern in analytical amounts using such tedious approaches.
This problem can now be solved using in vitro glycoengineering (IVGE). Since it can be applied on therapeutic proteins during downstream processing, the technology is independent from the cell line and the bioprocess used. Historically, this approach was not widely pursued because glycosyltransferases, the essential reagents for IVGE, were not available in sufficient amounts and in good quality. Recently, Roche Diagnostics together with Roche Pharma has developed sialyltransferases and a galactosyltransferase that now enable the biopharma industry to use IVGE to substantially improve the glycosylation profiles of therapeutic proteins.
This application note describes the assessment for suitability of the in vitro glycoengineering technology for the development, manufacturing, and analysis of therapeutic proteins.
We consider the following parameters to be most relevant for evaluation:
- Enzyme activity
- Implementation into the manufacturing process
- Enzyme versatility
- Growth in scale and quality during drug development
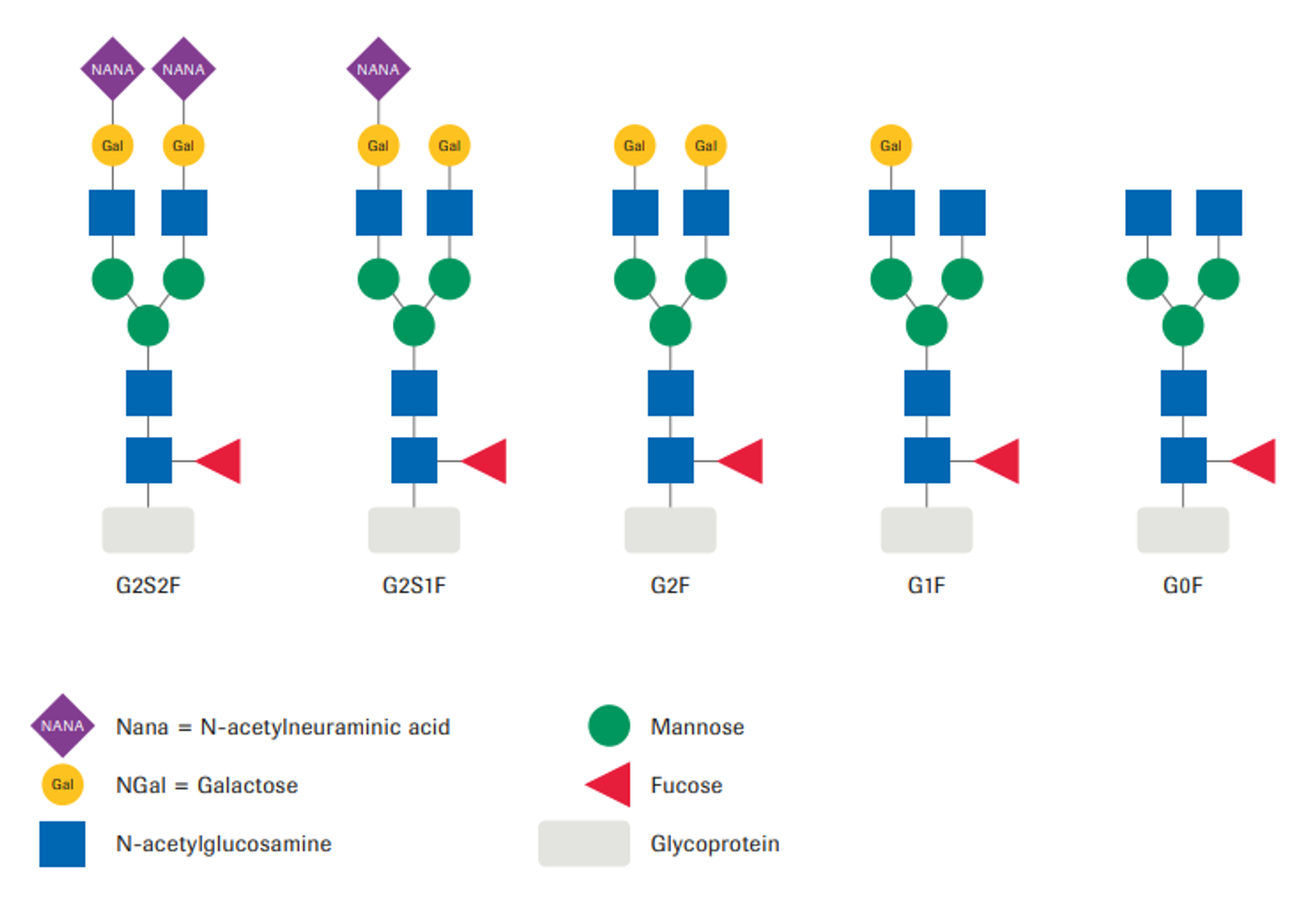
Glycan nomenclature
Enzyme activity of glycosyltransferases
The optimal reaction conditions for glycosylation of a target protein can be different for each application. Therefore, the presented protocols serve as a starting point. Optimization of reaction conditions (e.g., with different enzyme-IgG ratios) might be beneficial for each application.
Galactosylation using β-1,4-Galactosyltransferase, GalT1
Method
One milligram IgG1 was galactosylated according to the
protocol in the appendix. To monitor the progress of glycosylation, aliquots were taken at different time points and the degree of galactosylation was analyzed with mass spectrometry.
Result
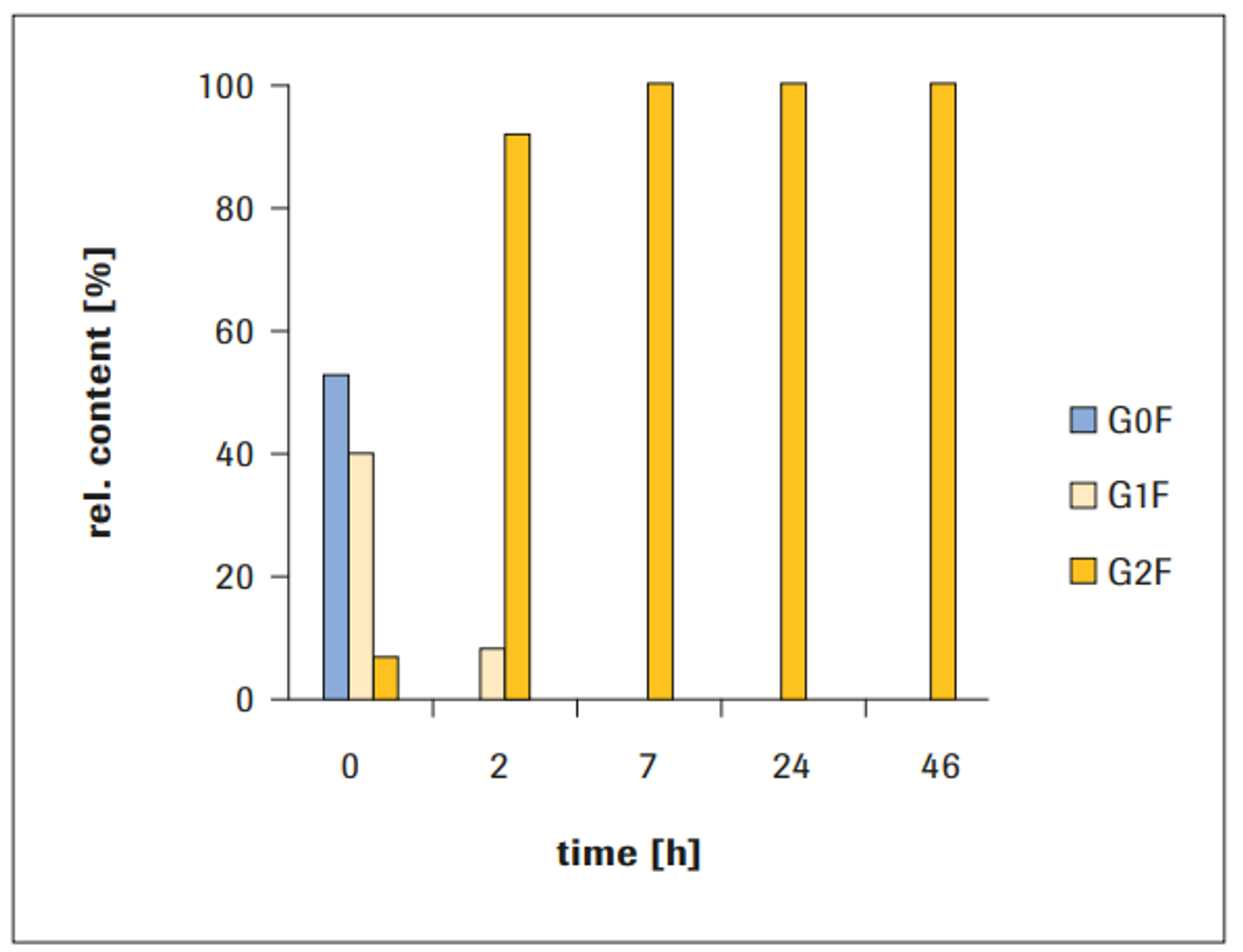
The starting material exhibited a heterogeneous glycosylation pattern. Species with no (G0F) and one galactose (G1F) were most abundant, whereas the bi-galactosylated variant (G2F) had a relative amount of less than 10%.
Full bi-antennary galactosylation was accomplished after 7 hours reaction time (Figure 1).
After 2 hours incubation, the main species was the G2F glycan with a relative amount of more than 90%.
Sialylation using α-2,3-Sialyltransferase, ST3
Method
One milligram IgG1 was sialylated according to the protocol
in the appendix. To monitor the progress of glycosylation, aliquots were taken at different time points and the degree of sialylation was analyzed using mass spectrometry.
Result
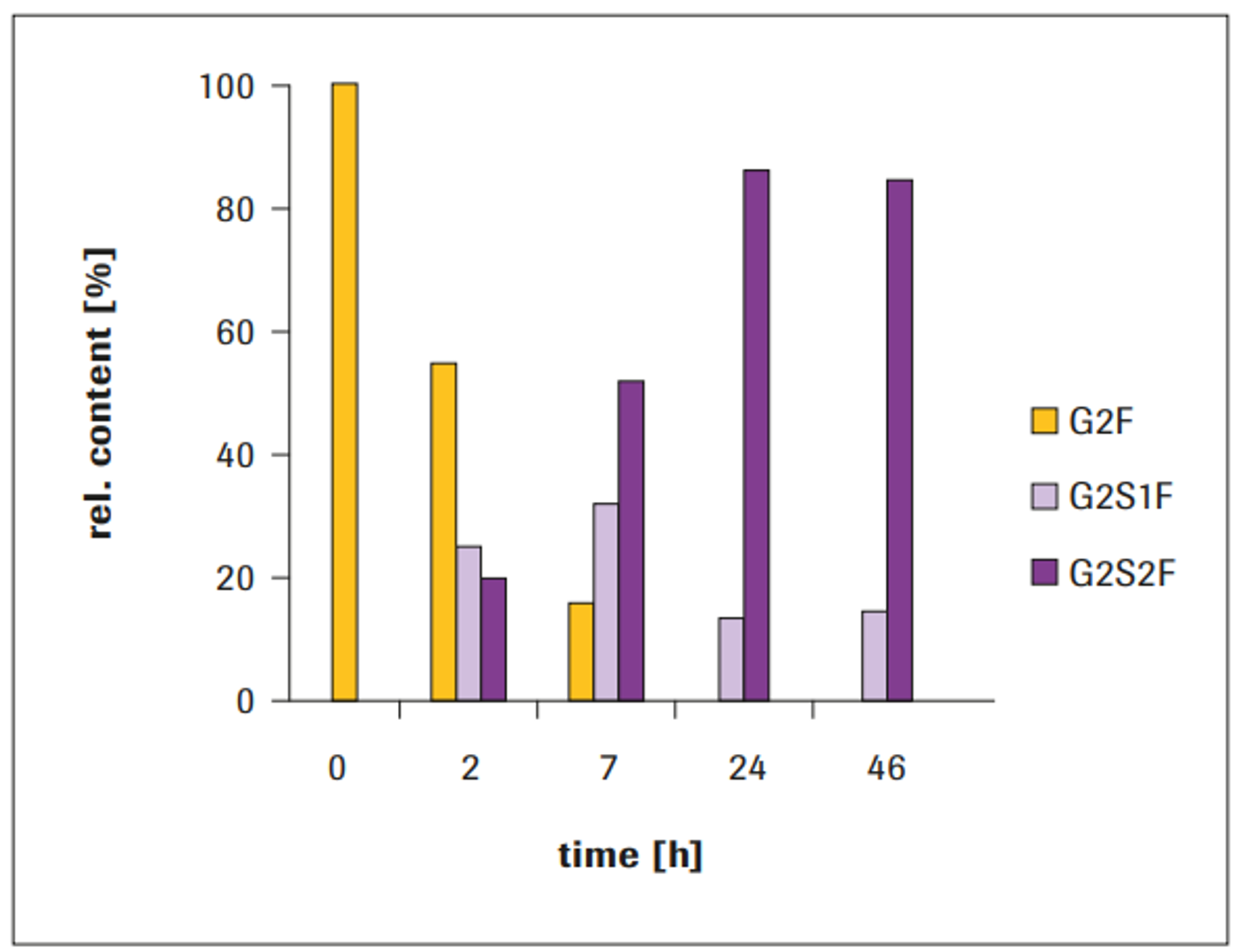
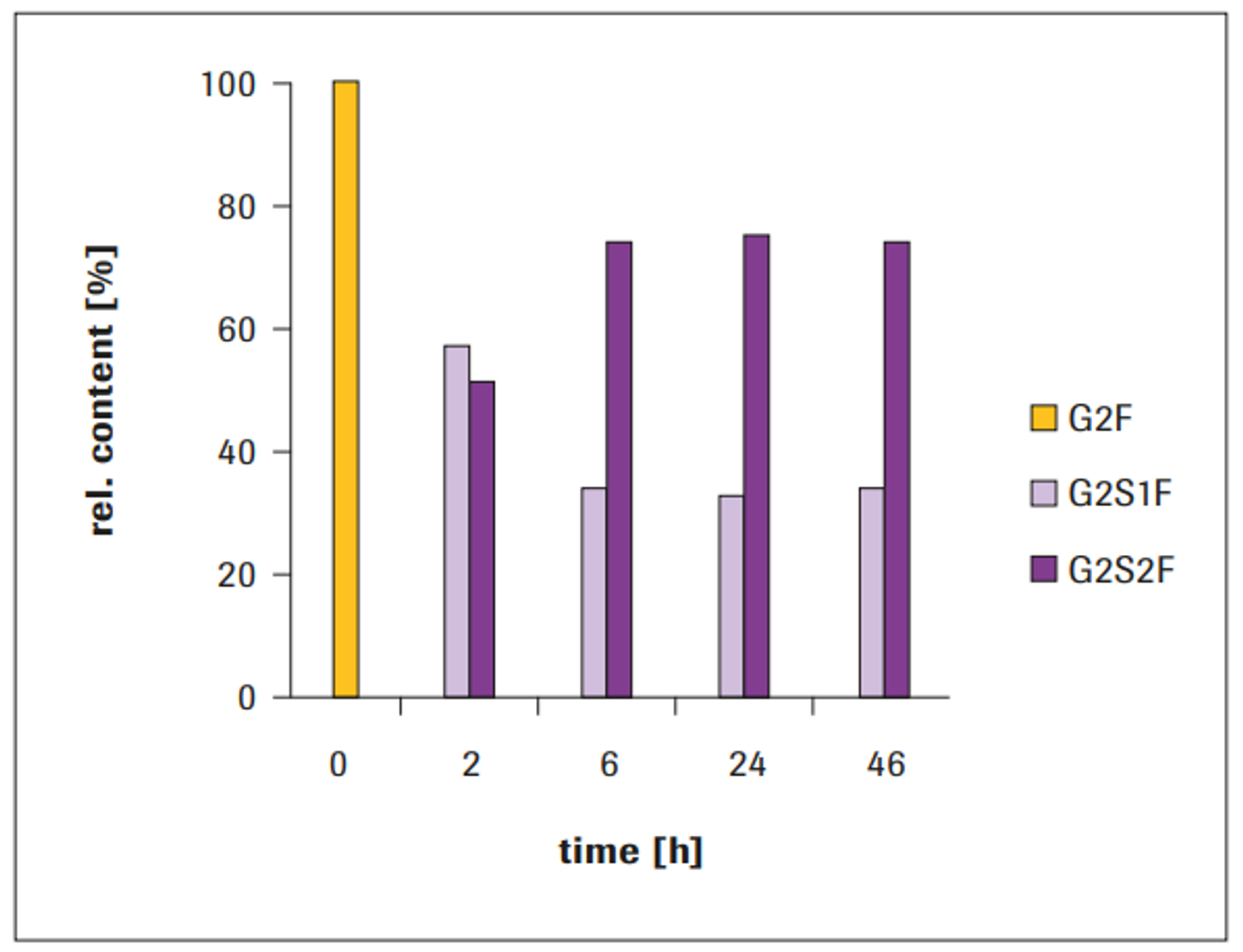
After 24 hours, a relative amount of approximately 85% G2S2F and approximately 15% G2S1F were achieved, which remained constant even after 46 hours incubation (Figure 2). Completely galactosylated IgG1 was used as acceptor molecule. After 2 hours, the glycan compostition comprised 25% G2S1F and 20% G2S2F. The sialic acid content showed further increase during incubation time, until a stationary state was reached after approximately 24 hours. Without addition of Alkaline Phosphatase (AP) to the reaction, a decrease in sialylation was observed at incubation times beyond approximately 7 hours (not shown).
Figure 1: Time course of IgG1 galactosylation (10 μg GalT1/mg IgG1)
Figure 2: Time course of IgG1 sialylation with ST3 (100 μg ST3/mg IgG1)
Sialylation using α-2,6-Sialyltransferase, ST6
Method
One milligram IgG1 was sialylated according to the protocol in the appendix. The progress of sialylation was analyzed using mass spectrometry. Completely galactosylated IgG was used as acceptor molecule.
Result
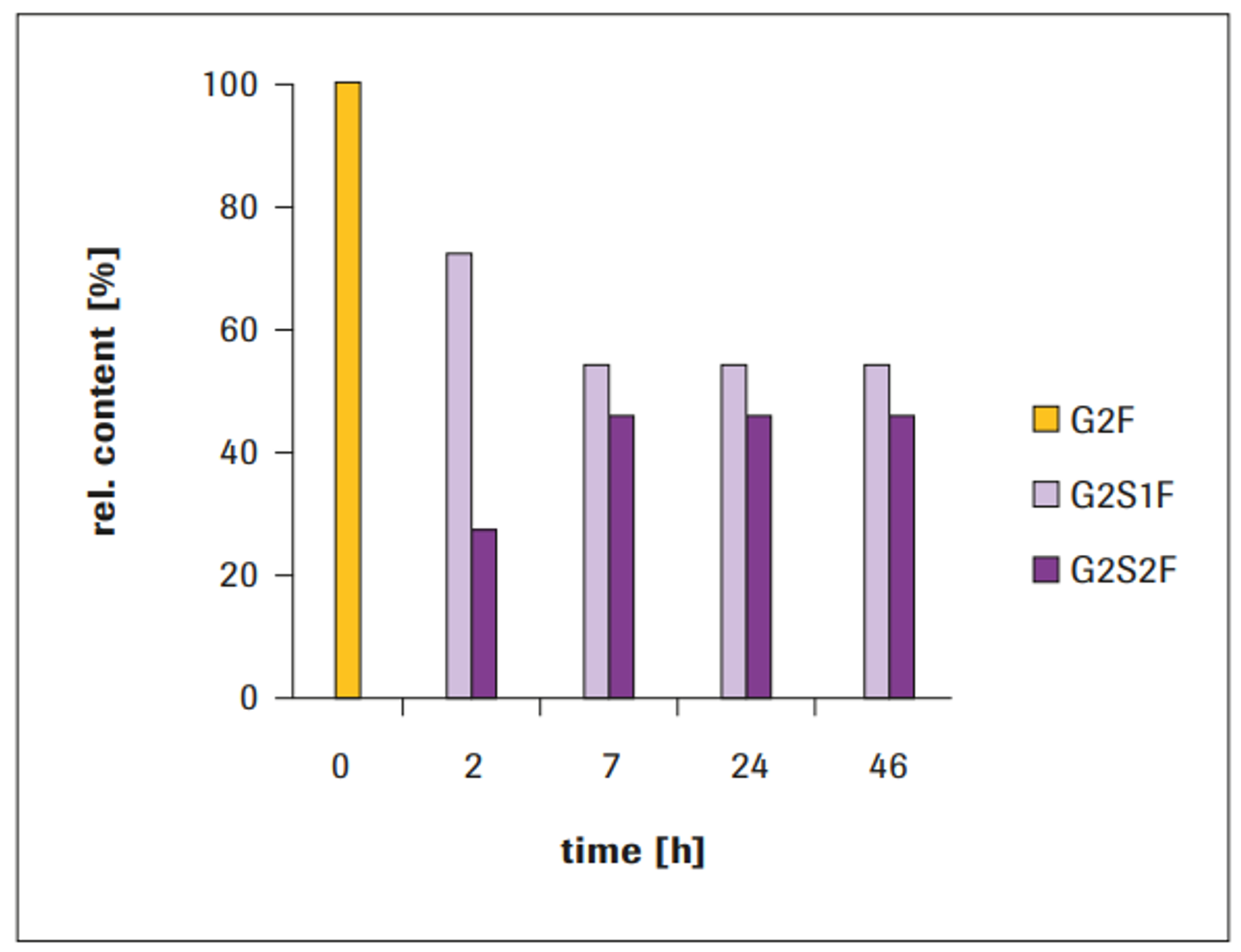
After 2 hours, the glycan composition comprised 72% G2S1F and 28% G2S2F. The bi-antennary sialylation revealed further increase during incubation time, up to a stationary state. After approximately 7 hours incubation, the maximum level of sialylation was reached: 46% G2S2F and 54% G2S1F, which remained constant even after 46 hours incubation (Figure 3). Without addition of AP to the reaction, a decrease in sialylati- on was observed at incubation times beyond approximately 7 hours (data not shown).
The sialylation using ST3 or ST6 was observed as very reliable and robust, even in slightly different buffer systems, provided that the pH of the reaction mixture was between pH 6 and 7: The data in Figure 3 were obtained from MES-buffered reactions. However, reactions in less ionic strength can even lead to increased maximal sialylation levels of up to 70% G2S2F (Figure 4).
Figure 3: Time course of IgG1 sialylation with ST6 (100 μg ST6/mg IgG1)
Figure 4: Improved sialylation if CMP-NANA is dissolved in water, instead of MES buffer (100µg ST6 /mg IgG1)
Implementation into the manufacturing process
Since the final drug substance must be free of glycosyltransferases, reaction buffer, and activated sugars, it would be advantageous if the IVGE step could be applied at an early downstream process intermediate, to avoid repetition of downstream process steps.
A second important aspect with regard to costs and time, especially in large-scale applications, is the possibility to perform both IVGE steps in a one-pot process.
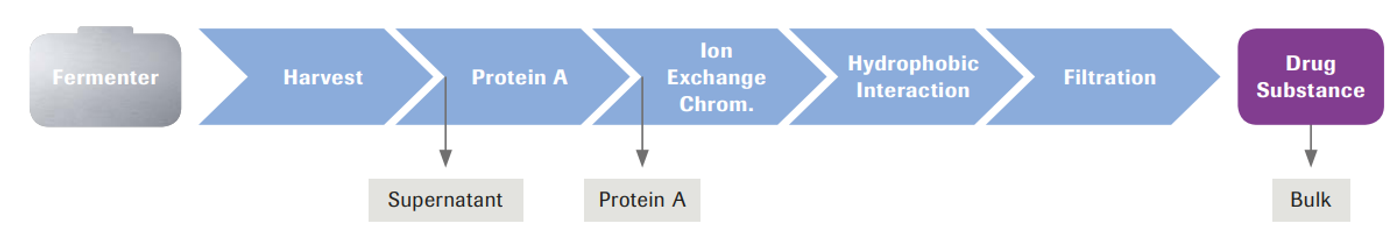
To address both questions, we used IVGE in a simultaneous galactosylation and sialylation reaction on IgG1 from different in-process steps: fermentation supernatant, Protein A eluate, and highly purified bulk (Figure 5).
Figure 5: Application of IVGE using different downstream intermediates, compared to purified bulk.
Simultaneous galactosylation and sialylation (“1-pot-reaction”)
Method
Bulk material, Protein A eluate, and fermentation supernatant, respectively, containing 1 mg IgG1 each, were galactosylated and sialylated without a purification step in between (protocol, see appendix). The progress of glycosylation was monitored by taking aliquots at different time points and subsequent analysis using mass spectrometry.
Result
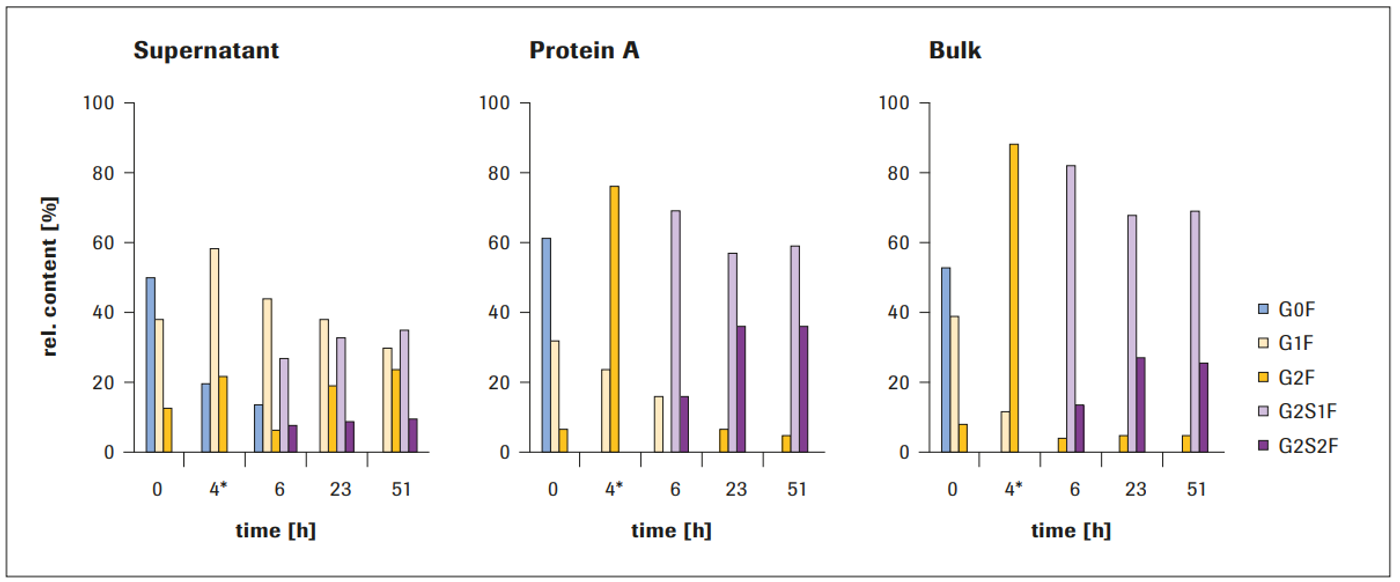
In all three samples, maximal bi-antennary sialylation was reached after 23 hours (Figure 6). The maximum level of sialylation in the 1-pot process was approximately 30% G2S2F and 60% G2S1F for both the Protein A and the bulk sample.
The levels were much lower for the supernatant sample. Therefore, use of Protein A-purified material or bulk material is recommended.
Figure 6: Results of IVGE using different downstream process intermediates (*incubation with GalT1 for 4 hours, followed by addition of ST6, CMP-NANA, and AP)
Enzyme versatility
All three enzymes (GalT1, ST3, ST6) showed comparably high activity on IgG1 and IgG4. They were also used to successfully glycosylateother recombinant glycoproteins (data not shown).
Growth in scale and quality during drug development
In contrast to research and analytical applications, the application of IVGE in the development of a commercial drug requires that the raw materials and the technologies grow together throughout the project, both in scale and with regulatory standards.
The glycosyltransferases meet the following important requirements:
- Scalability: The enzymes can be provided up to kg amounts upon request.
- Quality: Enzymes can be provided in GMP grade upon request.
- Animal-free status: No animal-derived materials were used in fermentation, purification, and final formulation of all three glycosyltransferases.
Summary and outlook – application of IVGE in biopharma industry
In vitro glycoengineering has already been used to produce small to medium amounts (up to 1 gram) of material for different analytical purposes. For example, IVGE was used to support structure-function analysis of early research projects (e.g., investigation of mode-of-action)2, pre-clinical studies (e.g., pharmacokinetic studies), comparability studies or CQA assessment of late-stage projects. The developed enzymes have successfully been tested on different therapeutic proteins, such as IgG1 and IgG4 antibodies.
The in vitro glycoengineering step can be integrated into existing downstream processes as shown for Protein A-purified IgG1.
The glycosyltransferases and activated sugars can be produced in GMP quality and animal free in large-scale amounts upon request to allow for modification of clinical material. Also, residual enzyme tests based on ELISA format are in development, which enables monitoring of successful removal of the enzymes after IVGE of clinical material. In our view, the prerequisites for an application in routine manufacturing should be fulfilled. The feasibility to employ the IVGE process in manufacturing scale needs to be tested and optimized for each application.
Appendix
Galactosylation of IgG
- Preparation of the reaction buffer, containing 10 mM UDP-Gal, 5 mM Manganese(II) Chloride (MnCl2), and 100 mM MES, pH 6.5.
- Dissolve 61 mg UDP-Gal, 6.3 mg MnCl2, and 195 mg MES in 6 ml water.
- Adjust pH to 6.5, and adjust to a final volume of 10 ml with water.
- Setting up the galactosylation reaction, containing 976 μg UDP-Galactose, 10 μg GalT1, and 1 mg IgG.
- 160 μl reaction buffer
- 33.3 μl of IgG (30 mg/ml, pH 6)
- 1.8 μl Galactosyltransferase
- 4.9 μl water
- Incubate the reaction at 37°C, then take aliquots and stop the reaction by freezing the tube at –15 to –25°C for mass spectrometry analysis.
Sialylation of IgG using either ST6 or ST3
- Preparation of the reaction buffer, containing 6.1 mM CMP-NANA, 200 mM MES, pH6.5.
- Dissolve 40 mg CMP-NANA and 390.4 mg MES in 6 ml of water.
- Adjust pH to 6.5, and adjust to a final volume of 10 ml with water.
- Setting up the sialylation reaction, containing 500 μg CMP-NANA, 100 μg Sialyltransferase, and 1 mg IgG.
- 125 μl reaction buffer
- 100 μl IgG (10 mg/ml, pH 6)
- 18 μl Sialyltransferase (ST3 or ST6)
- 2.8 μl Alkaline Phosphatase (1 mg/ml)
- 2.53 μl ZnCl2 solution (10 mM)
- 1.67 μl water
- Incubate the reaction at 37°C, then take aliquots and stop the reaction by freezing the tube at –15 to –25°C for mass spectrometry analysis.
Simultaneous galactosylation and sialylation (“1-pot-reaction”)
- Setting up the galactosylation reaction, containing 976 μg UDP-Galactose, 10 μg GalT1, and 1 mg IgG.
- 160 μl reaction buffer
- 33.3 μl of IgG (30 mg/ml, pH 6)
- 1.8 μl Galactosyltransferase
- 4.9 μl water
- Incubate the reaction for 4 hours at 37°C, then take 50 μl of this reaction for mass spectrometry analysis.
- Add reagents for subsequent sialylation to the remaining 150 μl galactosylated IgG, to set up the sialylation reaction, containing 375 μg CMP-NANA, 75 μg Sialyltransferase, and 0.75 mg IgG.
- 94 μl reaction buffer
- 13.6 μl Sialyltransferase ST6
- 2.9 μl Alkaline Phosphatase (1 mg/ml)
- 2.6 μl ZnCl2 solution (10 mM)
- Incubate the reaction at 37°C, then take aliquots and stop the reaction by freezing the tube at –15 to –25°C for analysis.
Note: In case of varying IgG concentrations, the protocol must be adapted. The most important parameter to be considered is the mass ratio of activated sugar, IgG and glycosyltransferase.
References
- Post-translational modifications in the context of therapeutic proteins, Gary Walsh & Roy Jefferis, Nat Biotechnol. 2006 Oct;24(10):1241-52
- In vitro glycoengineering of IgG1 and its effect on Fc receptor binding and ADCC activity, M. Thomann et al., PLoS One. 2015 Aug 12;10(8):e0134949
Regulatory disclaimer
α-2,6-Sialyltransferase, α-2,3-Sialyltransferase, β-1,4-Galactosyltransferase, Alkaline Phosphatase are: For life science research only. Not for use in diagnostic procedures. Activated sugars and glycosidases are: For further processing only.
License limitations
The sale of the product does not exhaust or grant any rights in third party patents including patents of companies of the F. Hoffmann - La Roche AG group of companies, in particular, for the use of modified antibodies obtained by using the product.