Discovery of Compound Provides New Hope for Treating Autoimmune Disease
The majority of drugs available for people with autoimmune diseases are only designed to alleviate symptoms, as opposed to targeting the root cause of disease. Thanks to the discovery of a new “drug-like compound,” University of Colorado at Boulder scientists believe they can work toward the development of new drugs that target the cause instead of just the symptoms.
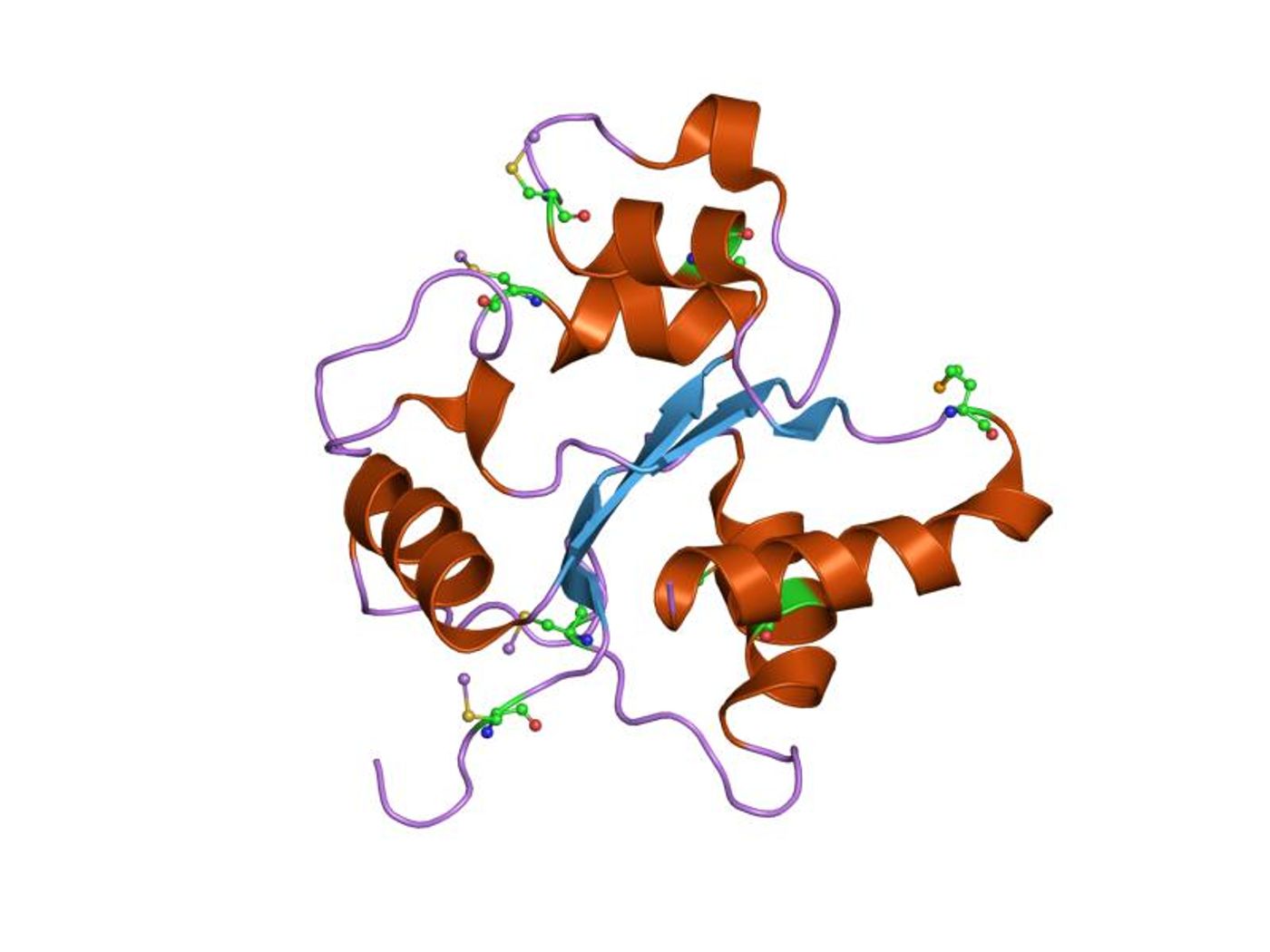
The new discovery starts with a component of the innate, or non-specific, immune system called toll-like receptor 8 (TLR8). Upon sensation of a pathogen, TLR8 triggers a domino reaction that changes the receptor from a passive state to an active state. Activate TLR8 releases inflammatory signals that are good for fighting bacteria and viruses, but bad for healthy tissues if the response goes overboard.
Historically, TLR8 has been difficult to study and apply as a drug target because it is “hidden inside the endosome,” while most drug targets are on the cell’s surface. The new CU Boulder study, in an effort to find a molecule to bind TLR8, completed high-throughput screening of more than 14,000 small molecule compounds. Out of the thousands, researchers zeroed in on just four with the potential to have the proper chemical structure for the job.
Using these four small molecules as a model, researchers chemically synthesized their own compound to bind TLR8, preventing TLR8 activity instead of silencing an already-active TLR8. "Before, people were trying to close the open door to shut it down. We found the key to lock the door from the inside so it never opens," explained lead author Hang Hubert Yin.
Yin calls the new synthesized compound and the approach to blocking TLR8 activation “paradigm shifting.”
In the future, Yin and his team plan on continuing their studies of the new TLR8-binding compound and its potential to treat a variety of autoimmune diseases that cause pain, inflammation, and other chronic health issues. Existing drugs can be costly and cause side effects, so the new compound could be the answer for many scientists looking for alternatives.
Yin is planning animal studies and human clinical trials, to learn more about TLR8 and other toll-like receptors.
The present study was published in the journal Nature Chemical Biology.